Abstracts
Abstract
This research was carried out to study the antifungal properties of Medicago sativa L. against phytopathogenic fungus Rhizoctonia solani J.G. Kühn. Methanolic extract of M. sativa was estimated in vitro against the R. solani. M. sativa methanolic extract was found to be highly efficient at the concentration of 5% as it reduced 74% test fungus biomass. Test plant was further partitioned by five organic fractions namely, n-butanol, chloroform, ethyl acetate, acetone and n-hexane. These isolated fractions and synthetic fungicide control group were evaluated in vitro against R. solani. Results showed that ethyl acetate fraction and fungicide were most effectively to repress the growth of R. solani up to 61-67%. This ethyl acetate fraction was further carried out for analysis of GC-MS and nine bioactive agents were found. The most copious compounds were phytol (RT: 20.84), 1,2,3-Propanetriol, monoacetate (RT: 12.47), Z,Z-3, 15-Octadecadien-1-olacetate (RT: 21.75), Hexadecanoic acid, ethyl ester (RT: 19.72) and Benzene, nitro- (RT: 10.20). The present study can be concluded that M. sativa possesses antifungal constituents against R. solani.
Keywords:
- bioassay,
- in vitro,
- GC-MS,
- methanolic extract
Résumé
Cette recherche a été menée afin d’étudier les propriétés antifongiques de Medicago sativa L. contre le champignon phytopathogène Rhizoctonia solani J.G. Kühn. L’extrait méthanolique de M. sativa contre R. solani a été évalué in vitro. L’extrait méthanolique de M. sativa s’est avéré très efficace à une concentration de 5%, car il réduisait de 74% la biomasse du champignon à l’essai. L’extrait de plante a ensuite été divisé en cinq fractions organiques à savoir le n-butanol, le chloroforme, l’acétate d’éthyle, l’acétone et le n-hexane. Ces fractions isolées et le groupe témoin de fongicide synthétique ont été évalués in vitro contre R. solani. Les résultats ont montré que la fraction d’acétate d’éthyle et le fongicide étaient les plus efficaces pour réprimer la croissance de R. solani jusqu’à 61-67%. Cette fraction d’acétate d’éthyle a ensuite été analysée par CPG-SM et neuf agents bioactifs ont été trouvés. Les composés les plus abondants étaient le phytol (TR : 20,84), le 1,2,3-Propanetriol, monoacétate (TR : 12,47), Z,Z-3, 15-Octadécadien-1-olacétate (TR : 21,75), l’acide hexadécanoïque, ester (TR : 19,72) et le benzène, nitro- (TR : 10,20). La présente étude peut être conclue que M. sativa possède des constituants antifongiques contre R. solani.
Mots-clés :
- bioessai,
- in vitro,
- CPG-MS,
- extrait méthanolique
Article body
INTRODUCTION
Fungi are among the major crop disease agents. Pathogenic fungi employ a range of approaches to colonize crops and cause illness. Some fungi nourish on their host’s dead material (necrotrophs), while others colonize residual tissue (biotrophs) (Doehlemann et al. 2017).
Rhizoctonia solani J.G. Kühn. is a ubiquitous phyto-pathogenic fungus born in the soil. It causes massive output losses in number of major agriculturally vital crops (Paulitz et al. 2006; Verma 1996). R. solani is among the quite notorious plant pathogen that causes post-emergence and pre-emergence damping-off diseases on many essential crops (Heydari et al. 2004). Before the development of plant, seed decay and death of the radicals occurs within the soil, which is the most common form of pre-emergence damping-off (Heydari et al. 2007). R. solani damages plants by producing hyphal threads in their germinating sclerotia. The fungus could not produce any conidia or asexual spores. Thanatephorus cucumeris is perfect stage of this fungus which appears as a narrow, mildew-like structure on soil, above the leaves and ground line (Uppala and Zhou 2018).
Plant pathogens require management of plant diseases. It is mandatory because up to ten percent of the foodstuff is decayed due to these pathogens (Strange and Scott 2005). Chemical pesticides are used to enabling plant fungal diseases to be managed quickly. The use of such pesticides has, however, raised public suspicions in recent times. Because of their cytotoxic, phytotoxic, carcinogenic and persistent effects, their practices are increasingly limited (Bajwa et al. 2008).
Botanical alternatives are safer than synthetic chemicals in the environment. The antifungal activity from the plant extracts have been shown to be effective against plant pathogens and ecofriendly (Duru and Onyedineke 2010; Latha et al. 2009). As an active source for fungitoxic substances, plant secondary metabolites have inordinate potential. Numerous operations such as antifungals and antimicrobials have been demonstrated by compounds produced by plants such as hydroquinone and sesquiterpenes (cinnamodial, capsidiol), naphthoquinone (lapachol, jugllone), and alkaloids (berberine) (El-Khateeb et al. 2013).
Alfalfa and lucerne are the common names of the M. sativa. It belongs to Fabaceae pea family grown as a common forage crop in many countries. Many essential secondary metabolites (coumarines, isoflavones, naphthoquinones, alkaloids and saponins) are generated by alfalfa. Medicago spp. have reported numerous biological actions, such as haemolytic activities, nematocidal characteristics, insecticidal properties, antibacterial and antifungal influences on humans and plants pathogens (Sadowska et al. 2014).
The purpose of the current study was to discover antifungal compound/antifungal constituents through the use of M. sativa extract to suppress the growth of R. Solani.
MATERIALS AND METHODS
Test experimental material collection
Medicago sativa sample plant was obtained from the area of Kana Kacha, Lahore. The test plant was dried under sunlight after thorough washing under tap water and finely grinded to make the powder. Pure culture of target fungus R. solani was obtained from the Laboratory of Fungal Biotechnology, LCWU (Lahore College for Woman University), Lahore. Sub culturing of this culture was done ant it was preserved on 2% MEA (Malt Extract Agar) and refrigerated at 4 °C.
Antifungal activity assay
Twenty-gram dried entire test plant powder of M. sativa was immersed in 100 mL of MeOH (methanol) at room temperature (± 30 °C) for six days. The content was filtered via a sterilized muslin cloth after 6 days. After evaporating at room temperature, the 3 g final methanolic greasy mass of M. sativa was attained. Twenty percent stock solution was produced by adding 15 mL distilled water into 3 g sticky mass of M. sativa.
In 250 mL flask, malt extract (1.2 g) and agar (1.2 g) were inserted in 60 mL of DW (distilled water) to attain 2% MEA and autoclaved at 121 °C for 30 min. Five test plant material’s concentrations (1%, 2%, 3%, 4% and 5%) of methanolic extract were formed by combining 0.3, 0.6, 0.9, 1.2 and 1.5 mL of stock solution into 59.7, 59.4, 59.1, 58.8 and 58.5 mL of DW (distilled water), respectively. In this way, total volume (60 mL) for each concentration was made. No plant extract was applied to any control group. Vibramycin capsule (100 mg) was introduced to each concentration as well as in control flask to prevent bacterial contamination. Using sterilized cork borer, 5 mm mycelial disks were prepared from one-week-old fungal culture and putted in the middle of all Petri dishes. Each treatment was made in triplicate manner. For one week, the whole setup was incubated at 25 ± 2 °C (Jabeen and Javaid 2010). The fungal growth diameter was calculated in centimeters after one week of fungal growth. Percentage decrease in each colony diameter size was calculated by given formula:
Bioassays with fractions of organic solvents
Various organic solvents were used to fractionate the M. sativa methanolic extract. For this, 40 g of plant material powder was immersed into 200 mL of MeOH (methanol) for seven days to acquire a methanolic extract of 20%. The acquired extract was then put to evaporate. For partitioning the extract, five organic solvents including n-hexane (100 mL), chloroform (CHCl3) (100 mL), acetone (100 mL), ethyl acetate (EtOAc) (100 mL) and n-butanol (100 mL) were separated by using a separating funnel. Such samples were evaporated at room temperature to get their gummy masses (Bashir et al. 2019). For each separate fraction and Mancozeb (synthetic fungicide), 20% stock solution was formed. For all five organic fractions, three concentrations were made. Antibiotic (vibromycin capsules) of 50 mg was also used to prevent bacterial impurities at each concentration. In control treatments there was no plant extract. Three replicates were created for each concentration. In each flask, five-millimeter disk of R. solani was inserted (Hanif et al. 2017). Antifungal activity was detected after one week and findings had been noted down.
GC-MS analysis
Ethyl acetate fraction of methanolic extract of M. sativa was preceded in further bioassay to identify potential antifungal compounds. M. sativa plant material (50 g) was soaked in EtOAc (250 mL) and rapidly shaken for 48 hours on incubator shaker for the full incorporation of metabolites into the organic solvent. Nylon membrane filter paper of diameter 47 mm was used with 0.22 µm of pore size to purify this extract. The extracted filtrate was then utilized for GC-MS (Gas Chromatography-Mass Spectrometry).
The test plant was examined using a GC-MS QP chromatograph and BD-5 (30 m, 0.25 mm, 0.25 μm) capillary column was used for isolation by implementing the temperature of 50 °C, using He (helium) (99.999%) as carrier gas (mobile phase). Temperatures for the injector and detector were set at 200 °C and 250 °C correspondingly. The parameters of the mass detector were 70 eV ionization voltages with 55-950 Da m Z-1 mass scanning range. Via peak areas of the GC, the volatile compounds proportion was estimated. Retention times (RT), mass spectra and indices were compared with earlier provided literature data provided by NIST Library 2010 word software for qualitative evaluation (Waheed et al. 2016).
Statistical analysis
Entire record was collected to examine on Statistix 8.1 software by applying the LSD (least significant difference) test. All the data were analyzed at significance level of P ≤ 0.05.
RESULTS AND DISCUSSION
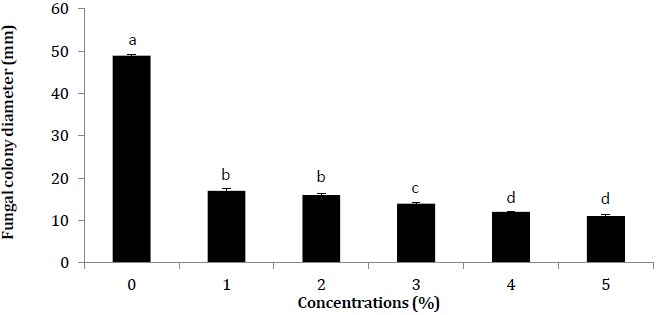
In present study, the Medicago sativa methanolic extract was tested against R. solani. Several methanolic extract concentrations (1% to 5%) of M. sativa inhibited R. solani development. The most efficient concentrations were 5% and 4%, since they restricted the R. solani growth up to 77% and 75% respectively (Fig. 1). Previously (Kagale et al. 2004), Datura metel’s antifungal efficacy was evaluated to retard growth R. solani and X. oryzae by using its methanolic and aqueous leaf extracts. Methanolic extract has been found to be considerably more efficient. The antifungal attributes are attributable to chemical substances formed by plants as phytochemicals such as polyphenols, which are involved in the mechanism for plants’ protection against bacteria and fungi (Castillo et al. 2010).
Figure 1
Impact of M. sativa plant methanolic extract on in vitro development of R. solani
Vertical bars display standard errors of means of 3 replicates. Alphabetic values at column show significant differences (P ≤ 0.05) as ascertained by software 8.1 Statistix.
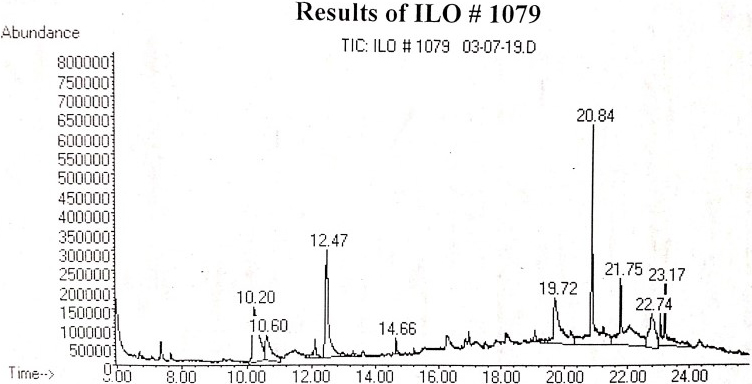
Figure 2
Impact of M. sativa extract of various concentrations on in vitro development of R. solani
Vertical bars display standard errors of means of 3 replicates. Alphabetic values at column show significant differences (P ≤ 0.05) as ascertained by software 8.1 Statistix.
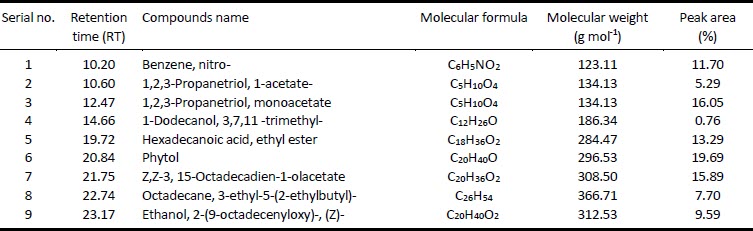
Figure 3
GC-MS chromatogram of the M. sativa ethyl acetate extract
Table 1
Compounds analyzed by GC-MS of M. sativa ethyl acetate extract
Partitioning of M. sativa methanolic extract was carried out using five organic fractions. The fraction of EtOAc (ethyl acetate) showed highest antifungal activity at concentrations of 1-2% as it suppressed fungal growth of 61-67% respectively. Treatment of control with synthetic fungicide also recorded 61-67% efficacy against test fungus R. solani at the concen-trations of 1-2%, respectively, the same as the fraction of ethyl acetate (Fig. 2). Other fractions also hindered growth of the test fungus. Previously, Seema et al. (2011) checked various organic solvent extracts of L. inermis, P. betel, P. longifolia and P. graveolens against R. solani and ethyl acetate fraction was registered the most substantial for antifungal activity testing against R. solani, as compared with other extracts.
The chromatograph of ethyl acetate extract of M. sativa revealed nine peaks of nine specific compounds after GC-MS analysis (Fig. 3). The leading constituents listed in the chromatogram of extract were phytol (RT: 20.84) 19.7%, 1,2,3-Propanetriol, monoacetate (RT: 12.47) 16.1%, Z,Z-3, 15-Octadecadien-1-olacetate (RT: 21.75) 15.9%, Hexadecanoic acid, ethyl ester (RT: 19.72) 13.3%, Benzene, nitro- (RT: 10.20) 11.7% and Ethanol, 2-(9-octadecenyloxy)-, (Z)- (RT: 23.17) 9.6% (Table 1). The compounds like Octadecane, 3-ethyl-5-(2-ethylbutyl)- (RT: 22.74) 7.7%, 1,2,3-Propanetriol, 1-acetate- (RT: 10.60) 5.3% and 1-Dodecanol, 3,7,11 -trimethyl- (RT: 14.66) 0.8% were present in lesser amount (Table 1). Earlier, Ponmathi et al. (2017) found the phytol in B. courtallica leaf extract which had shown notable antimicrobial activities. Previously, phytol recorded in Ipomoea staphylina ethanolic extract had shown great antimicrobial potential (Padmashree et al. 2018). Pejin et al. (2014) investigated the phytol’s anti-microbial assessment towards eight bacterial and fungal pathogens, and phytol was strong antimicrobial agent against all tested microorganisms. The earlier findings assisted our research work that the phytol present in EtOAc extract of Medicago sativa could have characteristic antimicrobial attributes. According to previous research, high concentrations of Benzene, nitro- had significant antifungal efficacy to retard the growths of M. Verrucaria and T. mentagrophytes (Gershon et al. 1971). Zekeya et al. (2014) founded that Hexadecanoic acid, methyl ester was present in root bark and Ethanol, 2-(9-octadecenyloxy)-, (Z)- was present in leaves of Bersama abyssinica methanolic extract which showed strong antimicrobial efficacy.
CONCLUSION
The current research came to the conclusion that M. sativa had substantial antifungal activity against R. solani and ethyl acetate fraction was found highly successful in combating target fungi and it might be due to the presence of various natural phytoconstituents examined in M. sativa via analysis of GC-MS.
Appendices
REFERENCES
- Bajwa, R., A. Javaid, S. Shafique, A. Javaid, K. Jabeen, and S. Shafique. 2008. Fungistatic activity of aqueous and organic solvent extracts of rice varieties on phytophathogenic fungi. Allelopathy J. 22: 363-370.
- Bashir, S., K. Jabeen, S. Iqbal, S. Javed, and A. Naeem. 2019.Lantana camara: Phytochemical analysis and antifungal prospective. Planta Daninha 37:1-7.
- Castillo, F., D. Hernández, G. Gallegos, M. Mendez, R. Rodríguez, A. Reyes, and C.N. Aguilar. 2010.In vitro antifungal activity of plant extracts obtained with alternative organic solvents against Rhizoctonia solani Kühn. Ind. Crop. Prod. 32: 324-328.
- Duru, C.M., and N.E. Onyedineke. 2010.In vitro study on the antimicrobial activity and phytochemical analysis of ethanolic extracts of the mesocarp of Voacanga africana. Amer. J. Plant Physiol. 5: 163-169.
- Doehlemann, G., B. Ökmen, W. Zhu, and A. Sharon. 2017. Plant pathogenic fungi. Microbiol. Spectr. 5: 1-23.
- El-Khateeb, A.Y., E.A. Elsherbiny, L.K. Tadros, S.M. Ali, and H.B. Hamed. 2013. Phytochemical analysis and antifungal activity of fruit leaves extracts on the mycelial growth of fungal plant pathogens. J. Plant Pathol. Microbiol. 4: 1-6.
- Gershon, H., M.W. McNeil, R. Parmegiani, and P.K. Godfrey. 1971. Antifungal activity of substituted nitrobenzenes and anilines. J. Appl. Microbiol. 22: 438-440.
- Hanif, S., K. Jabeen, and S. Iqbal. 2017. Management of damping off disease by extracts of Albizia lebbeck (L.) Benth. Bangl. J. Bot. 46: 1009-1014.
- Heydari, A., H. Fatahi, H. Zamanizadeh, Z.N. Hasan, and L. Naraghi. 2004. Investigation on the possibility of using bacterial antagonists for biological control of cotton seedling damping-off in green house. Appl. Entomol. Phytopathol. 72: 51-68.
- Heydari, A., I.J. Misaghi, and G.M. Balestra. 2007. Pre-emergence herbicides influence the efficacy of fungicides in controlling cotton seedling damping-off in the field. Int. J. Agric. Res. 2: 1049-1053.
- Jabeen, K., and A. Javaid. 2010. Antifungal activity of Syzygium cumini against Ascochyta rabiei, the cause of chickpea blight. Nat. Prod. Res. 24: 1158-1167.
- Kagale, S., T. Marimuthu, B. Thayumanavan, R. Nandakumar, and R. Samiyappan. 2004. Antimicrobial activity and induction of systemic resistance in rice by leaf extract of Datura metel against Rhizoctonia solani and Xanthomonas oryzae pv. oryzae. Physiol. Mol. Plant Pathol. 65: 91-100.
- Latha, P., T. Anand, N. Ragupathi, V. Prakasam, and R. Samiyappan. 2009. Antimicrobial activity of plant extracts and induction of systemic resistance in tomato plant by mixtures of PGPR strains and Zimmu leaf extract against Alternaria solani. Biol. Control 50: 85-93.
- Padmashree, M.S., R. Ashwathanarayana, R. Naika, and B. Roopa. 2018. Antioxidant, cytotoxic and nutritive properties of Ipomoea staphylina Roem & Schult. plant extracts with preliminary phytochemical and GCMS analysis. Asian J. Pharm. Pharmacol. 4: 473-492.
- Paulitz, T.C., P.A. Okubara, and W.F. Schillinger. 2006. First report of damping-off of canola caused by Rhizoctonia solani AG2-1 in Washington State. Plant Dis. 90: 829.
- Pejin, B., A. Savic, M. Sokovic, J. Glamoclija, A. Ciric, M. Nikolic, K. Radotic, and M. Mojovic. 2014. Further in vitro evaluation of antiradical and antimicrobial activities of phytol. Nat. Prod. Res. 28: 372-376.
- Ponmathi, S.A., E.R. Michael, S. Muthukumarasamy, and Vr. Mohan. 2017. Determination of bioactive components of Barleria courtallica nees (acanthaceae) by gas chromatography-mass spectrometry analysis. Asian J. Pharm. Clin. Res. 10: 273-283.
- Sadowska, B., A. Budzyńska, M. Więckowska-Szakiel, M. Paszkiewicz, A. Stochmal, B. Moniuszko-Szajwaj, M. Kowalczyk, and B. Różalska. 2014. New pharmacological properties of Medicago sativa and Saponaria officinalis saponin-rich fractions addressed to Candida albicans. J. Med. Microbiol. 63: 1076-1086.
- Seema, M., S.S. Sreenivas, N.D. Rekha, and N.S. Devaki. 2011.In vitro studies of some plant extracts against Rhizoctonia solani Kuhn infecting FCV tobacco in Karnataka Light Soil, Karnataka, India. J. Agric. Tech. 7: 1321-1329.
- Strange, R.N., and P.R. Scott. 2005. Plant diseases: a threat to global food security. Annu. Rev. Phytopathol. 43: 83-116.
- Uppala, S., and X.-G. Zhou. 2018. Rice sheath blight. Plant Health Instr. doi:10.1094/PHI-I-2018-0403-01.
- Verma, P.R. 1996. Biology and control of Rhizoctonia solani on rapeseed: a review. Phytoprotection 77: 99-111.
- Waheed, N., K. Jabeen, S. Iqbal, and A. Javaid. 2016. Biopesticidal activity of Calotropis procera L. against Macrophomina phaseolina. Afr. J. Tradit. Complement. Altern. Med. 13: 163-167.
- Zekeya, N., M. Chacha, F. Shahada, and A. Kidukuli. 2014. Analysis of phytochemical composition of Bersama abyssinica by gas chromatography-mass spectrometry. J. Pharmacogn. Phytochem. 3: 246-252.
List of figures
Figure 1
Impact of M. sativa plant methanolic extract on in vitro development of R. solani
Figure 2
Impact of M. sativa extract of various concentrations on in vitro development of R. solani
Figure 3
GC-MS chromatogram of the M. sativa ethyl acetate extract
List of tables
Table 1
Compounds analyzed by GC-MS of M. sativa ethyl acetate extract



 10.7202/706106ar
10.7202/706106ar