Abstracts
Abstract
Buckeye rot disease of tomato caused by Phytophthora nicotianae var. parasitica is the most destructive disease for reducing tomato yields especially in those regions where fruiting coincides with rainy season. In the present study, the pathogen was characterized by sequencing the DNA region coding for internal transcribed spacer (ITS) region and sequence was deposited in NCBI with accession no. MF398189. The phylogenetic analysis using the Maximum Composite Likelihood (MCL) approach revealed that the isolated pathogen clustered together with P. nicotianae with high bootstrap value of 99%. Incubation period of 120 h was observed in pin-prick method of pathogen inoculation compared to 168 h in surface inoculation method. Further, the disease resistance induced by nine different elicitors of induced resistance against buckeye rot disease of tomato were studied under field conditions for two consecutive years 2016 and 2017. Minimum disease incidence of 9.57% and 7.93% was observed with foliar spray of ß-aminobutyric acid (2 mM) for 2016 and 2017, respectively. It was followed by potassium chloride (100 mM) with disease incidence of 11.32% and 8.85% for year 2016 and 2017, respectively. Maximum fruit yield of 7.02 kg and 8.12 kg was found in treatment with ß-aminobutyric acid as compared to 2.61 kg and 2.55 kg in control for year 2016 and 2017, respectively.
Keywords:
- induced resistance,
- eco friendly,
- Phytophthora,
- inducers
Résumé
Le mildiou zoné de la tomate causé par Phytophthora nicotianae var. parasitica est la maladie la plus destructrice qui cause une réduction des rendements de tomates, en particulier dans les régions où la fructification coïncide avec la saison des pluies. Dans cette étude, l’agent pathogène a été caractérisé par séquençage de la région d’ADN codant pour la région de l’espaceur interne transcrit (ITS) et la séquence a été déposée sur NCBI avec le numéro d’accession MF398189. L’analyse phylogénétique utilisant l’approche de probabilité composite maximale (MCL) a révélé que l’agent pathogène isolé se regroupait avec P. nicotianae avec une valeur de bootstrap élevée à 99 %. Une période d’incubation de 120 h a été réalisée avec la méthode d’inoculation de l’agent pathogène par piqûres d’aiguilles comparativement à 168 h avec la méthode d’inoculation en surface. De plus, la résistance à la maladie induite par neuf éliciteurs différents contre le mildiou zoné de la tomate a été étudiée sur le terrain pendant deux années consécutives, en 2016 et en 2017. Une incidence minimale de la maladie de 9,57 % et de 7,93 % a été observée avec une pulvérisation foliaire d’acide ß-aminobutyrique (2 mM) pour 2016 et 2017 respectivement, suivi du chlorure de potassium (100 mM) avec une incidence de la maladie de 11,32 % et 8,85 % pour les années 2016 et 2017 respectivement. Un rendement maximal en fruits de 7,02 kg et de 8,12 kg a été trouvé dans le traitement avec de l’acide ß-aminobutyrique par rapport à 2,61 kg et 2,55 kg dans le traitement contrôle pour les années 2016 et 2017 respectivement.
Mots-clés :
- résistance induite,
- écologique,
- Phytophthora,
- éliciteurs
Article body
INTRODUCTION
Phytophthora species cause very destructive diseases in large number of plant species resulting in huge yield losses every year. Buckeye rot disease caused by Phytophthora nicotianae var. parasitica, considerably constrains the tomato production especially in hilly regions of India. Under high humidity and good rainfall conditions, the incidence of buckeye rot alone may go up to 90% (Gupta and Thind 2006). The situation is likely to worsen because the versatility on the development of resistance in Phytophthora population to fungicides with specific mode of action such as metalaxyl had been demons-trated in many reports. The fungicide metalaxyl targets RNA polymerase I thereby inhibiting the synthesis of ribosomal RNA in Phytophthora (Davidse et al. 1988). Since long time, isolates of Phytophthora infestans showing resistance against metalaxyl, an oomycete-specific fungicide, had been reported frequently from different parts of the world (Davidse et al. 1981; Deahl et al. 1993; Goodwin et al. 1996; Mukalazi et al. 2001; Iram and Ahmad 2009; Randall et al. 2014; Shattock 1988). Resistance of P. infestans to metalaxyl in the USA was first reported in 1993 in which none of 73 isolates of P. infestans were found sensitive to metalaxyl (Deahl et al. 1993). Among the 73 isolates, 81% that recovered from diseased potato tissues were highly resistant to 10 µg metalaxyl mL-1 whereas the rest (19%) showed intermediate resistance. The inheritance of resistance to metalaxyl is complex and may involve multiple genes. Recent research indicated that a single nucleotide polymorphism (SNP) in the gene encoding RPA190, the largest subunit of RNA polymerase I, confers resistance to metalaxyl (or mefenoxam) in some isolates of the potato late blight pathogen P. infestans (Matson et al. 2015).
The excessive use of pesticides to control plant diseases and development of resistance in pathogens is a problem for today’s plant production systems, and research priorities call for novel protection methods which are compatible with sustainable agriculture, thus favouring the use of alternative methods such as the application of chemical inducers or elicitors of resistance. Elicitors activate plants’ defence and result in induction of resistance. Elicitors of induced resistance such as salicylic acid application and pathogen-inoculation enhanced NPR1 (Nonexpressor of Pathogenesis Related Genes 1) expression and overexpression of ArabidopsisNPR1 or its homologs conferred broad resistance against diverse pathogens in multiple plant species (Potlakayala et al. 2007). NPR1 gene, also called NIM1 (Non-Inducible Immunity 1) or SAI1 (Salicylic Acid Insensitive 1) gene, is the key factor in the expression of induced resistance. NPR1 proteins are normally present in the cytoplasm and shuttle into the nucleus after monomerization due to changes in redox potential of the cell. NPR1 monomers interact with transcription factors and promoters of defence genes to establish induced resistance (Dong 2004; Sharma 2016). Application of elicitors had been reported to provide resistance in plants against diseases caused by Phytophthora spp. Soil drench and foliar spray of “Saver”, a product containing salicylic acid as active ingredient and β-aminobutyric acid (BABA), induced systemic acquired resistance against Phytophthora capsici and reduced the severity of P. capsici in squash (Koné et al. 2009). Eschen-Lippold et al. (2010) reported that systemic resistance was induced in potato plants against the devastating late blight pathogen P. infestans, with the treatment of BABA. BABA had been shown to move systemically in many plants including tomato, tobacco, and grape plants and provided the systemic protection against diseases in plants (Cohen 1993). BABA induces resistance against many foliar, fruit, root and soil pathogens and also against abiotic stresses (Cohen et al. 2016). BABA is a simple, 4-carbon, non-protein amino acid with the formula CH3-CH-(NH2)-CH2-COOH. However, the records on use of elicitors of induced resistance in tomato against P. nicotianae var. parasitica are not available yet. So, in this present investigation we have used nine different elicitors of induced resistance to evaluate their efficacy to induce resistance in tomato plants against P. nicotianae var. parasitica under field conditions and to provide an alternative management strategy for buckeye rot disease.
MATERIALS AND METHODS
Isolation and identification of pathogen
To isolate the pathogen from infected tomato fruits showing typical symptoms of buckeye rot, standard isolation procedure was performed on Petri plates containing carrot-juice agar medium (Dhingra and Sinclair 1995). Cultural and morphological characters, viz. colour and appearance of colony, size and colour of the hyphae, size and shape of sporangia were compared with standard description for the genus Phytophthora (Waterhouse 1963).
To ascertain the identity of the pathogen, genomic DNA was isolated from seven-day-old fungal cultures using Cetyl-tri-methyl ammonium bromide (CTAB) method (Murray and Thompson 1980). Quantity of isolated DNA was checked by gel electrophoresis on 0.8% of agarose gel. The DNA sequence coding for the internal transcribed spacer (ITS) region was amplified from the genomic DNA of the pathogen in the polymerase chain reaction (PCR) using universal primers ITS1 (5’-TCC GTA GGT GAA CCT GCG-3’) and ITS4 (5’-CTC CGC TTA TTG ATA TGC T-3’) 25 µL reaction volumes. PCR reaction volume contains 1.0 unit Taq DNA polymerase, dNTP mix (0.2 mM each of dCTP, dGTP, dATP and dTTP), 1 × PCR buffer (20 mM Tris–HCl pH 8.4, 50 mM KCl, 1.5 mM MgCl2), 1.0 µM each primer and 25-50 ng of genomic DNA. The PCR amplification was carried out in a thermocycler (Veriti, Applied Biosystems) with a total of 40 cycles. The PCR products were separated on 1.2% gel in horizontal electrophoresis (BIORad). The amplified PCR product on agarose gel was visualized under UV light using “Ingeneous” gel documentation system (Syngene). The PCR amplified product was eluted from the gel using GeneJET Gel Extraction Kit (Thermo Scientific) by following the manufacturer’s protocol. For DNA sequencing, purified PCR product was outsourced to Xcleris Genomics India under refrigerated conditions using gel packs and thermocol box. The sequence obtained was analyzed with the help of Bioinformatics tools like BioEdit, ClustalW and NCBI-BLAST (Basic Local Alignment Search Tool) for homology search. Phylogenetic analysis between sequence of isolated pathogen and the homologous sequences available in NCBI-BLAST was conducted in MEGA6 (Tamura et al. 2013). The evolutionary history was inferred by using the Maximum Likelihood method based on the Tamura 3-parameter model (Tamura 1992). The bootstrap consensus tree inferred from 1000 replicates was taken to represent the evolutionary history of the taxa analyzed (Felsenstein 1985). Branches corresponding to partitions reproduced in less than 50% bootstrap replicates were collapsed. Initial tree(s) for the heuristic search were obtained automatically by applying neighbour-join and BioNJ algorithms to a matrix of pairwise distances estimated using the Maximum Composite Likelihood (MCL) approach, and then selecting the topology with superior log likelihood value. The analysis involved 15 nucleotide sequences. All positions containing gaps and missing data were eliminated.
Pathogenicity of P. nicotianae var. parasitica
Preparation of sporangial suspension
To induce sporangial production, a Petri plate containing seven-day-old culture on carrot-juice agar medium was kept inverted on a plate of same size containing 20 mL of 0.01 M potassium nitrate solution prepared in sterilized distilled water under aseptic conditions and this pair of plates thus formed was sealed with adhesive tape to make it airtight. In order to accomplish the vapour condensation process for the induction of sporangial formation, the sealed pair was care-fully placed in an incubator at 30 °C for 30 min, subsequently removed and placed in a refrigerator at 5 ± 1 °C for 10 min and thereafter incubated at 25 ± 1 °C for 24 h. This process was repeated twice and after 72 h of vapour condensation process, the sporangia thus formed were harvested by flooding the culture plate with 10 mL of sterilized distilled water through two washings with the help of camel hair brush and the resulting suspension was filtered through muslin cloth to avoid mycelial fragments.
Pin-prick method of inoculation
Healthy green fruits of tomato were collected, thoroughly washed and disinfected with ethanol (70%). The disinfected fruits were then rinsed in four changes of sterilized distilled water and were dried in air before inoculation. The fruits were then pierced with sterilized needle at three places and 0.5 µL sporangial suspension (9 × 104 sporangia mL-1) of test pathogen was placed on the wounded portion of the fruit using micropipette, placed in sealed moist plastic box along with sterilized filter paper sprayed with sterilized water to maintain approximately 95 ± 5% relative humidity and was incubated for 10 days at 25 ± 1 °C.
Surface inoculation method of inoculation
Healthy green fruits of tomato were surface sterilized with ethanol (70%) and inoculated by placing a drop (5 µL) of sporangial suspension (9 × 104 sporangia mL-1) of test pathogen prepared in sterilized distilled water, with the help of micro-pipette on the surface of the fruits and was covered with a cotton swab. The inoculated fruits were incubated at 25 ± 1 °C and 95 ± 5% relative humidity for 10 days in plastic box along with sterilized filter paper sprayed with sterilized water.
Evaluation of resistance-inducers against buckeye rot of tomato
Nine different elicitors of induced resistance (Table 1) were evaluated for their efficacy to induce resistance against buckeye rot of tomato caused by P. nicotianae var. parasitica under field conditions for two consecutive years i.e., 2016 and 2017.
The efficacy of various elicitors of induced resistance was studied under field conditions during 2016 and 2017 crop season at the experimental farm of the Department of Plant Pathology, where disease incidence was recorded high in previous years. Experiment was conducted by using susceptible variety “Himsona”. The experiment was laid out in randomized block design. Each treatment was repeated thrice. Plot size was 2.0 × 2.0 m, having three rows at 60 cm distance was maintained. Five seedlings (25 day-old) were transplanted in each row at 30 cm spacing between the seedlings and total fifteen plants per plot were transplanted. Foliar spray of elicitors was applied on grown-up plants after 25 days of transplanting and repeated once after 15 days of first spray. The observations on disease incidence were recorded at a weekly interval till completion of crop season. The data on the yield of fruits were also recorded on each harvesting periodically. The disease incidence (DI) and reduction in disease incidence were calculated as described above.
Statistical analysis
The ANOVA of disease incidence readings were made after transforming the data of each replication with the following formula:
RESULTS AND DISCUSSION
Identification of pathogen
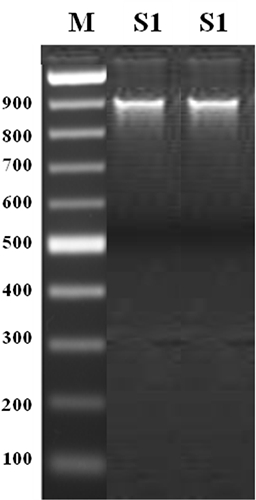
The mycelium of isolated fungus was coenocytic, hyaline, slender, 4.5–9.0 µm diam, profusely branched, with granular protoplasm, fluffy and aerial. Sporangiophores arose from hyphal thread and produced sporangia terminally and intercalary. Sporangiophores were hyaline, narrow, slender, irregularly branched, indeterminate, aseptate with sparse thick walls, side branches having bulbous enlargements at intervals. The sporangia were terminal, ovoid to globose, papillate measuring about 22-58 × 20-45 µm in size. The pathogen was identified as P. nicotianae (Breda de Haan) var. parasitica (Dastur) Waterhouse (Waterhouse 1963). P. nicotianae var. parasitica is characterized by no swellings on hypha, hyphal diameter up to 9 µm, papillate sporangia broadly ovoid or ellipsoid to spherical formed on irregularly branched sympodial sporangiophores and spherical or occasionally ellipsoid sporangia that are caducous with short pedicels, chlamydo-spores forming late (10 to 14 days) and usually about 22 to 30 µm diam with thicker walls (3 to 4 µm) (Waterhouse 1963, 1974). Whereas P. nicotianae var. nicotianae is characterized by hyphae up to 5 µm diam with frequent hyphal swellings, spherical to obturbinate, noncaducous, papillate sporangia formed terminally on the sporangiophore and not in a sympodium, abundant chlamydospores (20 to 40 µm diam) with walls about 1.5 µm thick (Waterhouse 1963, 1974; Waterhouse and Waterston 1964). Additionally, P. parasitica was characterized by papillate sporangia produced singly or in a loose sympodium on long stalks, chlamydospores are terminal or intercalary and 13 to 60 µm diam (average 28 µm) (Hall 1993; Thomson and Hine 1972). To confirm the identity of Phytophthora sp., DNA sequence coding for ITS region was amplified and a unique band of 900 bp was obtained (Fig. 1). NCBI-BLAST search revealed that closest sequence was of P. nicotianae. The sequence of the P. nicotianae of the present study was submitted to National Centre for Biotechnology Information (NCBI) and its accession number is MF398189.
The phylogenetic analysis using the MCL approach revealed that the isolate S1 with NCBI accession no. MF398189 clustered together with P. nicotianae with high bootstrap value of 99% (Fig. 2). The phylogram was clearly divided into two clusters: Cluster I consisted of Phytophthora spp. and cluster II grouped Pythium spp. Cluster I was further subdivided into Ia, Ib and Ic. Ia consisted of S1 isolate (accession no. MF398189) and P. nicotianae (accession no. KT148938.1). Subcluster Ib grouped isolates of P. nicotianae and P. parasitica. Subcluster Ic consisted of P. capsici only.
Table 1
Evaluation of elicitors of induced resistance against buckeye rot of tomato under field conditions during 2016 and 2017
Values in parentheses are arcsine transformed. * Critical Difference (CD) or Least Significant Difference (LSD)
Figure 1
Amplification of genomic DNA of Phytopthora nicotianae var. parasitica isolate S1 with primer pair ITS1/ITS4 for DNA region coding for internal transcribed spacer (ITS)
Pathogenicity and incubation period of P. nicotianae var. parasitica causing buckeye rot of tomato
Tomato fruits were inoculated with sporangial suspension (9 × 104 sporangia mL-1) by following either pin-prick method or surface inoculation method. Incubation period of 120 h was observed in pin-prick method compared to 168 h incubation period in surface inoculation method. Hence, prick method was found to be superior over the surface inoculation method for proving the pathogenicity of the P. nicotianae var. parasitica (Fig. 3). Shah (2009) while proving pathogenicity of P. nicotianae var. parasitica reported that maximum incidence (78.66%) of buckeye rot was observed in injured tomato fruits inoculated with culture bits than in uninjured fruits inoculated with culture bits on the surface with 60.45% of disease incidence. The appearance of symptoms was observed for 10 days. The symptoms produced after inoculation were found to be identical to symptoms observed under natural conditions as described in literature (Sharma and Singh 1992; Shridhar et al. 2018). The pathogen was reisolated from the infected fruit on carrot-juice agar medium. The microscopic characters of reisolated pathogen were same as recorded in the original culture of the test pathogen and colony characteristics of both the cultures were same. This proved the Koch’s postulates and pathogenicity of the isolated pathogen.
Figure 2
Phylogenetic analysis of DNA sequence coding for internal transcribed spacer (ITS) region of Phytopthora nicotianae var. parasitica isolate S1 with NCBI accession no. MF398189 through Maximum Likelihood method with bootstrap analysis
Figure 3
Pathogenicity of Phytophthora nicotianae var. parasitica on tomato fruits by pin-prick method (a, b) and surface inoculation method (c, d)
(a, c): Un-inoculated control showing healthy green fruits. (b, d): Development of symptoms of buckeye rot disease on tomato fruits after pathogen inoculation.
Field efficacy of elicitors of resistance against buckeye rot of tomato under field conditions
The results from the foliar spray of elicitors of induced resistance under field conditions revealed that ß-aminobutyric acid (2 mM) was most effective in inducing the disease resistance against buckeye rot of tomato with 86.86% reduction in disease incidence (Table 1). Minimum disease incidence of 9.57% and 7.93% was found with foliar spray of ß-amino-butyric acid for years 2016 and 2017, respectively. It was followed by potassium chloride (100 mM) with 11.32% and 8.85% disease incidence compared to 67.49% and 65.72% disease incidence in control for years 2016 and 2017, respectively. Foliar spray of oxalic acid (20 mM), salicylic acid (20 mM), sodium salicylate (10 mM) and potassium oxalate (50 mM) were next best in order with 19.31%, 25.54%, 29.05% and 30.39% disease incidence, respectively. Foliar spray of dipotassium hydrogen phosphate (50 mM) and silicic acid (0.002%) were least effective in inducing the disease resistance with 44.50% and 50.49% disease incidence, respectively. All the treatments significantly increased the overall yield of harvested fruits as compared to control. Maximum yield of fruits i.e., 7.58 kg per plot was obtained from the ß-aminobutyric acid treated plants followed by potassium chloride and oxalic acid with 7.47 and 6.89 kg per plot of fruit yield, respectively. Application of different elicitors, viz. ß-aminobutyric acid, potassium chloride, oxalic acid, salicylic acid, sodium salicylate and potassium oxalate were very effective to reduce the incidence of buckeye rot disease with 86.86%, 84.85%, 71.01%, 61.66%, 56.39% and 54.38% reduction in disease incidence, respectively. These elicitors were comparable in their efficacy for reduction of disease with commonly used fungicides against buckeye rot of tomato. Reduction up to 90% and 84% in incidence of buckeye rot disease of tomato was reported with fungicide metalaxyl + mancozeb and cymoxanil + mancozeb, respectively (Gupta and Bharat 2008). Three systemic new generation fungi-cides, viz. fenamidone + mancozeb, famoxadone + cymoxanil and metiram + pyraclostrobin were also effective to reduce the disease with 75.2%, 70.1% and 69.6% reduction in disease incidence under natural epiphytotic conditions, respectively (Shridhar et al. 2018).
Field evaluation of efficacy of different elicitors of induced resistance, viz. ß-aminobutyric acid, potassium chloride and oxalic acid showed their effectiveness against the buckeye rot disease of tomato caused by P. nicotianae var. parasitica. Plant diseases caused by Phytophthora species are usually difficult to manage. Foliar sprays with fungicides and varietal resistance are recommended as control measures of the disease. Elicitors of induced resistance provide an additional option to manage buckeye rot disease while maintaining sustainable production. Application of elicitors of induced resistance through foliar spray during present investigations represents a novel approach to manage buckeye rot of tomato.
The non-protein amino acid ß-aminobutyric acid had been shown to induce resistance in Arabidopsis against P. parasitica through activation of defence mechanisms such as callose deposition, hypersensitive response and the formation of trailing necrosis which inhibit the growth of the pathogen (Jakab et al. 2001). The spray application of ß-aminobutyric acid resulting in induced resistance against different species of Phytophthora had been mentioned in literature by earlier workers (Cohen 1993; Gilardi et al. 2014; Koné et al. 2009).
The inhibitory effect of potassium ions against plant pathogens has also been reported by many workers. Potassium ions resulted in the collapse of hyphal walls and inhibition of sporangial production of P. cinnamomic and P. drechsleri (Halsall and Forrester 1997). El-Mohamedy et al. (2013) also reported that potassium salts i.e., potassium sorbate and potassium carbonate completely inhibited the mycelial growth of Phytophthora spp. (isolate Ph MPC) at concentration of 8% and 4%, respectively. Hence, the efficacy of potassium chloride against buckeye rot disease can be attributed to its ability to inhibit sporangial production in Phytophthora spp. The rapid absorption of potash by the plant tissues and their extreme mobility within tissues as well as their low cost and their nutrient value make them ideal fertilizers besides controlling the disease.
The overall result of the present investigations demons-trated that treatment of tomato plants with elicitors of induced resistance developed an induced resistance against P. nicotianae var. parasitica infection. Foliar sprays of elicitors of resistance could provide an environmentally safe alternative to chemical fungicides for the management of buckeye rot to tomato. Two foliar sprays of elicitors of induced resistance namely ß-aminobutyric acid, potassium chloride and oxalic acid were effective for management of buckeye rot disease and could become potential disease management agents against diseases caused by Phytophthora spp. in a wide array of cultivated crops.
Appendices
REFERENCES
- Cohen, Y. 1993. Local and systemic control of Phytophthora infestans in tomato plants by DL-3-amino-n-butanoic acids. Phytopathology 84: 55-59.
- Cohen, Y., M. Vaknin, and B. Mauch-Mani. 2016. BABA-induced resistance: milestones along a 55-year journey. Phytoparasitica 44: 513-538. doi:10.1007/s12600-016-05 46-x
- Davidse, L.C., O.C.M. Gerritsma, J. Ideler, K. Pie, and G.C.M. Velthuis. 1988. Antifungal modes of action of metalaxyl, cyprofuram, benalaxyl and oxadixyl in phenylamide-sensitive and phenylamide-resistant strains of Phytophthoramegasperma f. sp. medicaginis and Phytophthora infestans. Crop Prot. 7: 347-355. doi:10.1016/0261-2194(88)90001-4
- Davidse, L.C., D. Looijen, L.J. Turkensteen, and D. van der Wal. 1981. Occurrence of metalaxyl-resistant strains of Phytophthora infestans in Dutch potato fields. Neth. J. Plant Pathol. 87: 65-68.
- Deahl, K.L., D.A. Inglis, and S.P. DeMuth. 1993. Testing for resistance to metalaxyl in Phytophthora infestans isolates from northwestern Washington. Am. Potato J. 70: 779-795.
- Dhingra, O.D, and J.B. Sinclair. 1995. Basic plant pathology methods. CRC Press. 448 pp.
- Dong, X. 2004. NPR1, all things considered. Curr. Opin. Plant. Biol. 7: 547-552.
- El-Mohamedy, R.S.R., M.M. Abdel-Kader, F. Abd-El-Kareem, and N.S. El-Mougy. 2013. Essential oils, inorganic acids and potassium salts as control measures against the growth of tomato root rot pathogens in vitro. Int. J. Agric. Technol. 9: 1507-1520.
- Eschen-Lippold, E., S. Altmann, and S. Rosahl. 2010. DL-beta-aminobutyric acid-induced resistance of potato against Phytophthora infestans requires salicylic acid but not oxylipins. Mol. Plant Microbe Interact. 23: 585-592.
- Felsenstein, J. 1985. Confidence limits on phylogenies: an approach using the bootstrap. Evolution 39:783-791.
- Gilardi, G., S. Demarchi, M. Gullino, and A. Garibaldi. 2014. Managing Phytophthora crown and root rot on tomato by pre-plant treatments with biocontrol agents, resistance inducers, organic and mineral fertilizers under nursery conditions. Phytopathol. Mediterr. 53: 205-215. doi:10. 14601/Phytopathol_Mediterr-12361
- Goodwin, S.B., L.S. Sujkowski, and W.E. Fry. 1996. Widespread distribution and probable origin of resistance to metalaxyl in clonal genotypes of Phytophthora infestans in the United States and Western Canada. Phytopatholy 86: 793-800.
- Gupta, S.K., and N.K. Bharat. 2008. Management of buckeye rot and late blight of tomato through combi-fungicides. Pestology 32: 17-19.
- Gupta, S.K., and T.S. Thind. 2006. Disease problems in vegetable production. Scientific Publishers, Jodhpur, India. 576 pp.
- Hall, G. 1993. An integrated approach to the analysis of variation in Phytophthora nicotianae and a redescription of the species. Mycol. Res. 97: 559-574. doi:10.1016/S0953-7562(09)81179-9
- Halsall, D.M., and R.I. Forrester. 1997. Effects of certain cations on the formation and infectivity of Phytophthora zoospores. 1. Effects of calcium, magnesium, potassium, and iron ions. Can. J. Microbiol. 23: 994-1001. doi:10. 1139/m77-148
- Iram, S., and I. Ahmad. 2009. A review of metalaxyl resistance in Phytophthora infestans from Pakistan. Acta Hortic. 834: 155-160.
- Jakab, G., V. Cottier, V. Toquin, G. Rigoli, L. Zimmerli, J.-P. Metraux, and B. Mauch-Mani. 2001. ß-aminobutyric acid-induced resistance in plants. Eur. J. Plant Pathol. 107: 29-37.
- Koné, D., A.S. Csinos, K.L. Jackson, and P. Ji. 2009. Evaluation of systemic acquired resistance inducers for control of Phytophthora capsici on squash. Crop Prot. 28: 533-538.
- Matson, M.E.H., I.M. Small, W.E. Fry, and H.S. Judelson. 2015. Metalaxyl resistance in Phytophthora infestans: assessing role of RPA190 gene and diversity within clonal lineages. Phytopatholy 105: 1594-1600. doi:10.1094/PHY TO-05-15-0129-R
- Mukalazi, J., E. Adipala, T. Sengooba, J.J. Hakiza, M. Olanya, and H.M. Kidanemariam. 2001. Metalaxyl resistance, mating type and pathogenicity of Phytophthora infestans in Uganda. Crop Prot. 20: 379-388. doi:10.1016/S0261-2194(00)00145-9
- Murray, M.G., and W.F. Thompson. 1980. Rapid isolation of high molecular weight plant DNA. Nucleic Acids Res. 8: 4321-4325.
- Potlakayala, S.D., C. DeLong, A. Sharpe, and P.R. Fobert. 2007. Conservation of non-expressor of pathogenesis-related genes1 function between Arabidopsis thaliana and Brassica napus. Physiol. Mol. Plant Pathol. 71: 174-183.
- Randall, E., V. Young, H. Sierotzki, G. Scalliet, P.R.J. Birch, D.E.L. Cooke, M. Csukai, and S.C. Whisson. 2014. Sequence diversity in the large subunit of RNA polymerase I contributes to Mefenoxam insensitivity in Phytophthora infestans. Mol. Plant Pathol. 15: 664-676. doi:10.1111/m pp.12124
- Shah, T.A. 2009. Cause and management of Phytophthora fruit rot of tomato (Lycopersicon esculentum Mill.) in Kashmir. Sher-e-Kashmir University of Agricultural Sciences and Technology of Kashmir, India. 224 pp.
- Sharma, M. 2016. Potential of induced resistance to manage diseases of vegetable crops. Pages 255-270 in P. Chowdappa, P. Sharma, D. Singh, and A.K. Misra (eds.), Perspectives of plant pathology in genomic era. Today & Tomorrows Printers and Publishers, New Delhi, India.
- Sharma, R.C., and H. Singh. 1992. Occurrence of Phytophthora nicotianae var. parasitica on hybrid tomato in Punjab. Plant Dis. Res. 7: 90-91.
- Shattock, R.C. 1988. Studies on the inheritance of resistance to metalaxyl in Phytophthora infestans. Plant Pathol. 37: 4-11.
- Shridhar, B.P., M. Sharma, S.K. Gupta, and S.K. Sharma. 2018. New generation fungicides for the management of buckeye rot of tomato. Indian Phytopathol. 71: 621-625.
- Tamura, K. 1992. Estimation of the number of nucleotide substitutions when there are strong transition-transversion and G+C-content biases. Mol. Biol. Evol. 9: 678-687. doi:10.1093/oxfordjournals.molbev.a040752
- Tamura, K., G. Stecher, D. Peterson, A. Filipski, and S. Kumar. 2013. MEGA6: Molecular Evolutionary Genetics Analysis version 6.0. Mol. Biol. Evol. 30: 2725-2729.
- Thomson, S.V., and R.B. Hine. 1972. Atypical sporangium-like structures of Phytophthora parasitica. Mycologia 64: 457-460.
- Waterhouse, G.M. 1963. Key to the species of Phytophthora de Bary. Mycological papers no. 92. Commonwealth Mycological Institute, Kew, England. 22 pp.
- Waterhouse, G.M. 1974.Phytophthora palmivora and some related species. Pages 51-70 in P.H. Gregory (ed.), Phytophthora disease of cocoa. Longman, London, England. 336 pp.
- Waterhouse, G.M., and J.M. Waterston. 1964.Phytophthora nicotianae var parasitica. CMI Descriptions of Pathogenic Fungi and Bacteria. No. 33. 2 pp.
List of figures
Figure 1
Amplification of genomic DNA of Phytopthora nicotianae var. parasitica isolate S1 with primer pair ITS1/ITS4 for DNA region coding for internal transcribed spacer (ITS)
Figure 2
Phylogenetic analysis of DNA sequence coding for internal transcribed spacer (ITS) region of Phytopthora nicotianae var. parasitica isolate S1 with NCBI accession no. MF398189 through Maximum Likelihood method with bootstrap analysis
Figure 3
Pathogenicity of Phytophthora nicotianae var. parasitica on tomato fruits by pin-prick method (a, b) and surface inoculation method (c, d)
List of tables
Table 1
Evaluation of elicitors of induced resistance against buckeye rot of tomato under field conditions during 2016 and 2017